|
The two-year, phase I/IIa study will enroll a total of 12 participants aged 18 years and older, with a body mass index of less than 35. 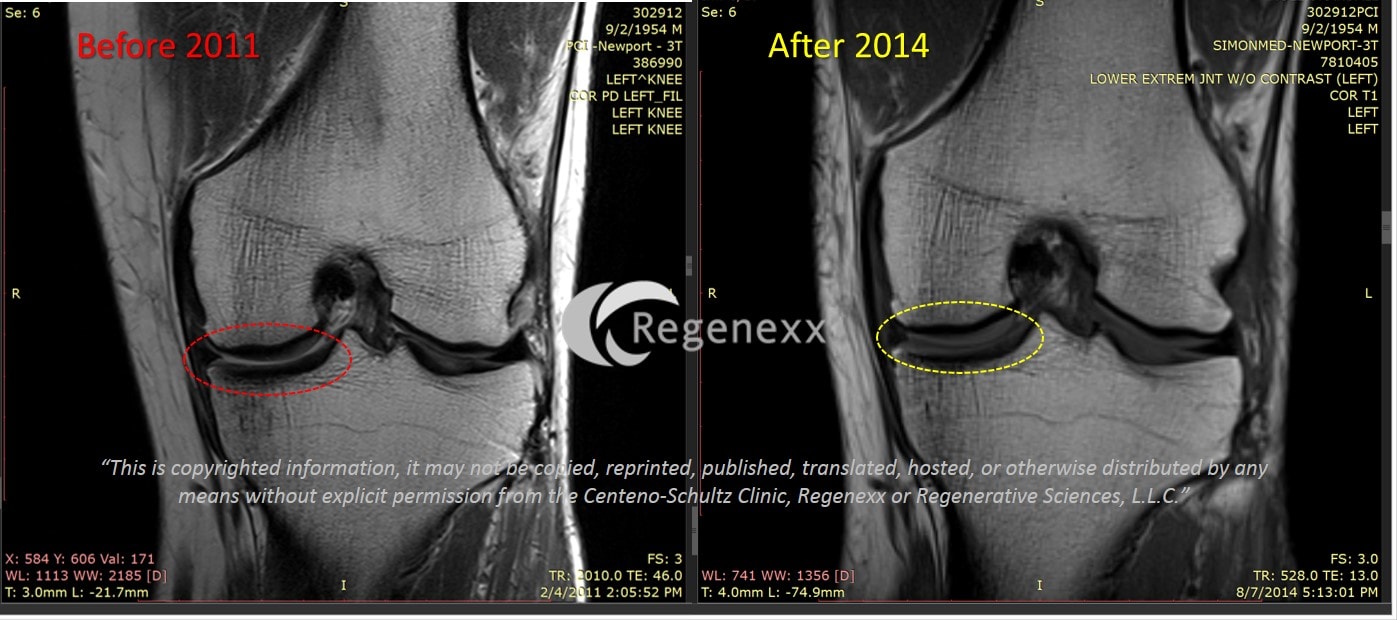
"Rush is spearheading this field of research with the ultimate goal of safely improving outcomes and sparing patients from having more complicated surgery at a relatively young age." "Finding a biological solution for cartilage regeneration in orthopedics is one of the fastest growing areas of research and development in our specialty, said Cole. Various treatments for cartilage degeneration, such as drug therapy, arthroscopy and joint replacement, yield mixed results and are unable to regenerate damaged tissue. 
Treating cartilage damage can be problematic because the tissue does not contain blood vessels or nerves and therefore has a limited ability to re-grow. Cole and his co-researchers will assess the drug's safety as well as its ability to regenerate cartilage repair tissue and reduce pain in patients with localized cartilage loss in the knee. Cole is the head of Rush's Cartilage Restoration Center and is also the head team physician for the Chicago Bulls. Brian Cole, a professor in the department of orthopedics and anatomy and cell biology at Rush University Medical Center. The principal investigator on the study is Dr. Cartistem is surgically administered into the area of cartilage damage following an arthroscopic surgery as an adjunct to microfracture, a commonly used technique used to repair cartilage damage. The stem cells are mixed with hyaluronan, a natural polymer that plays a major role in wound healing and is a building block of joint cartilage. Umbilical cord blood is a readily accessible source of high-quality stem cells, is associated with minimal health risks and carries relatively few ethical concerns. Cartistem is manufactured from mesenchymal stem cells derived from allogeneic (donor) umbilical cord blood.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
